 
Unfortunately, there was a slightly different system in place in Europe. There are seven periods in the periodic table, with each one beginning at the far left. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. A period is a horizontal row of the periodic table. They are organized in a tabular format, where a row represents a period, and a column represents a group. 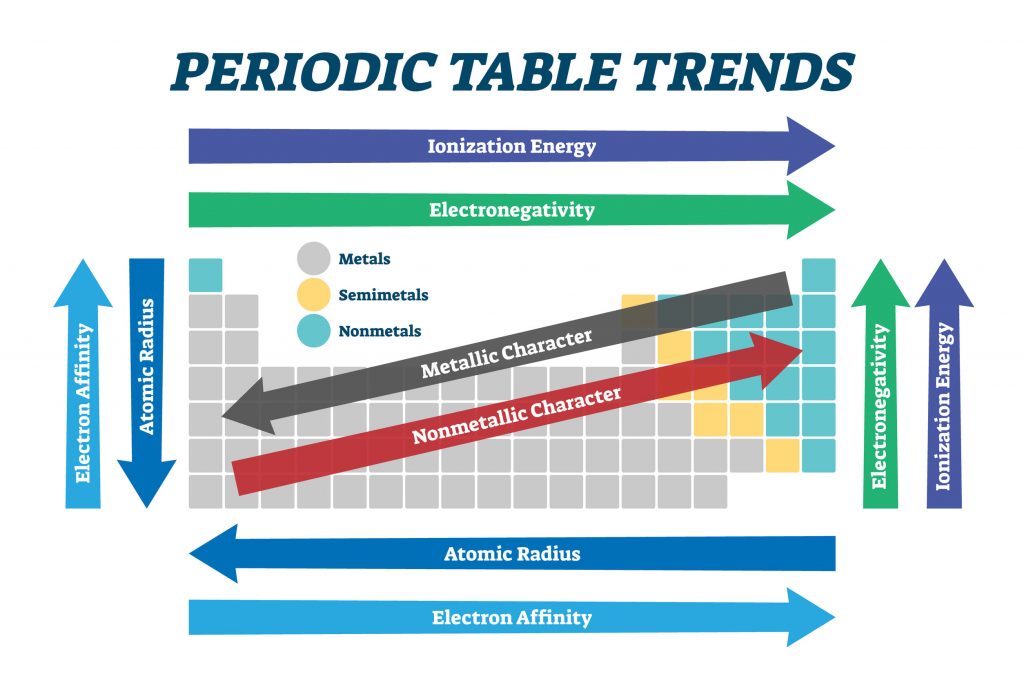
The traditional system used in the United States involves the use of the letters A and B. The periodic table is an arrangement of all known elements in order of increasing atomic number and recurring chemical properties. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These observed patterns allude to the changes in atomic structure, including size and radius, as well as properties of the elements. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periodic trends are specific patterns observed among the chemical elements of the periodic table. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. 
Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. It was not until 1911 that Rutherford conducted his gold foil experiment that demonstrated the presence of the nucleus in the atom. This tells us the number of atoms per unit volume of the solid. Ionic Radius - The ionic radius is a measure of the size of the spherical region around the nucleus of an ion within which the electrons are most likely to be found. When Mendeleev put his periodic table together, nobody knew about the existence of the nucleus. A rough idea of the size of a metallic atom can be obtained simply by measuring the density of a sample of the metal. The radii of atoms and ions are typically in the range 70-400 pm. There are seven periods in the periodic table, with each one beginning at the far left. 1 pm 10 12 m 10 10 cm 10 2 Å, or 1Å 100 pm. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
